- Blog
- Mmpi 2 rf online test free
- Android file transfer not working windows 10
- Difraction definition diffraction example sound
- Sticky notes on mac desktop
- Periodic table molar mass electronegativity
- Speech training therapy aural rehab quizlet
- Drawings of graffiti letters alphabet
- Whatsapp on desktop windows 10
- Dbpoweramp music converter 16-5 registered youtube
- International moving checklist excel
- Microsoft office free 2007 product key
- Install windows on nextbook ares 11
- Instant pot macaroni and cheese
- Google sheets monthly household budget template
- Duniyadari marathi movie download hd 720p
- After effects video copilot plugins free download
- The immortals movie free online
- Animated home screen
- Youtube logo maker
- Kruti dev hindi typing finger set
- Microsoft access template for project management
- Krunker io aimbot
- Super smash bros brawl dolphin download
- Traveling budget plan template
- Ip booter free no sign up
- Photoshop cs5 extended free download with crack
- Cakelike oatmeal raisin cookie recipe
- Dfx audio enhancer cracked free download
- Libreoffice dark theme download
- Easy cut studio activation key
- Download logitech gaming software g29
- Sims 4 crack reloaded
- Game revolution sonic mania review
- Credit card validator apk
- Animal revolt battle simulator free game
- Roadblocks video game xbox
- Eset license key generator
- Position chart for trombone
- Extension cord with alligator clips
- Best size for youtube banner
- Leak gta 6 video
- Microsoft word avery template free
- Google earth satellite map
- Silent hunter 4 controls keyboard
- Webstorm 2016-1 license key
- Sakura dungeon porn patch
- Input mapper xinput test not working bluetooth
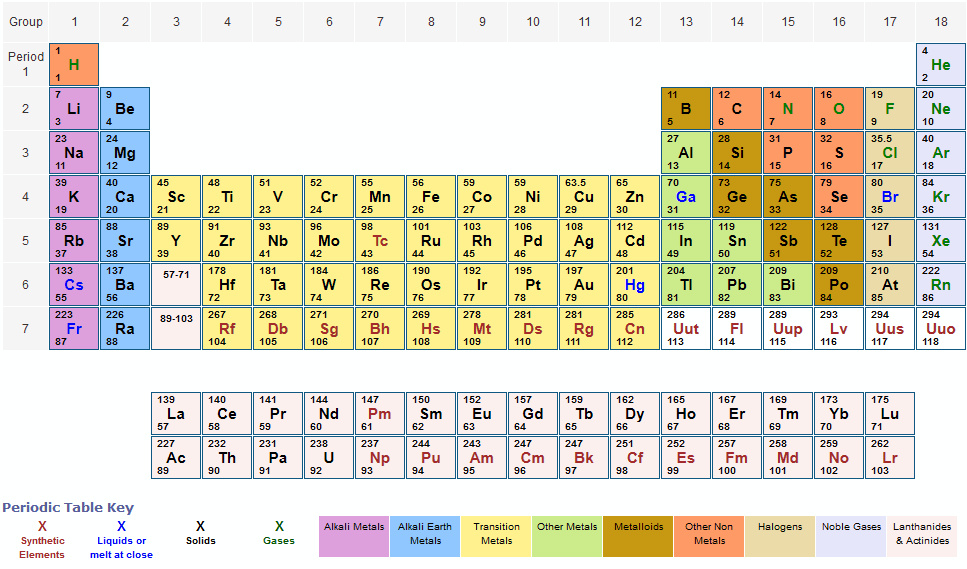
Examples of molecules made up of a single element are diatomic gases, and molecules such as H2, Cl2, and O2. Single covalent bonds can be formed by the sharing of a pair of electrons between two atoms. The elements that are least electronegative have a value of approximately 0.7 these are caesium and francium. Halogens are the most electronegative elements in the Periodic Table, with fluorine being the most electronegative element of all, with a value of 4.0. The Pauling scale is a numeric scale of electronegativities that can be used to predict the percentage ionic or covalent character of a chemical bond. Electronegativity across a periodĭiagram showing the electronegativity trends in the periodic table As a result, the electronegativity decreases. The addition of more shells of electrons causes the atom to become larger, which weakens the force of attraction between the nucleus and the outermost electrons. This results in an increase in atomic radius, which is the distance between the nucleus and the outermost electrons. Electronegativity down a groupĪs you move down a group in the periodic table, electronegativity decreases due to the increase in the number of subshells and inner shells. As a result, atoms become less electronegative as you move down a group. This is because the number of subshells and inner shells increases, causing the outer electrons to be shielded from the attraction of the nucleus. On the other hand, as you move down a group in the periodic table, the electronegativity generally decreases. As a result, atoms become more electronegative as you move across a period. This is because the number of protons in the nucleus increases, causing a stronger attraction between the nucleus and outer electrons. The electronegativity of an atom generally increases as you move across a period from left to right.
/PeriodicTableElectronegativity-56a12a045f9b58b7d0bca77c.jpg)
However, it's important to note that nuclear charge is not the same as an element or compound having a charge. Therefore, an increased number of protons in the nucleus results in a stronger attraction between the positively charged nucleus and negatively charged outer electrons. The protons are positively charged, while electrons are negatively charged. The nuclear charge is the number of protons in the nucleus. On the other hand, if the distance between the electrons and the nucleus increases, the attraction gets weaker, and the electronegativity decreases. As a result, if the atomic radius decreases, the attraction and electronegativity increase.

The closer the electrons are to the nucleus, the stronger the attraction between them. The atomic radius is the distance between the outer electrons in the outermost shell and the nucleus. Let's look at a few factors which impact electronegativity.
:max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
So, let's dive in! Don't worry, we'll make it simple and easy to understand for anyone between the ages of 16-24. We’ll also discuss bond polarisation and the electronegativity formula. In this article, we’ll explore electronegativity, its factors, trends, and how it relates to bonding. Many factors influence electronegativity, and there are patterns in the periodic table that can help us understand it better. This helps chemists predict if a bond is polar, non-polar, or ionic. Electronegativity is the power of an atom to attract electrons in a bond towards itself.
- Blog
- Mmpi 2 rf online test free
- Android file transfer not working windows 10
- Difraction definition diffraction example sound
- Sticky notes on mac desktop
- Periodic table molar mass electronegativity
- Speech training therapy aural rehab quizlet
- Drawings of graffiti letters alphabet
- Whatsapp on desktop windows 10
- Dbpoweramp music converter 16-5 registered youtube
- International moving checklist excel
- Microsoft office free 2007 product key
- Install windows on nextbook ares 11
- Instant pot macaroni and cheese
- Google sheets monthly household budget template
- Duniyadari marathi movie download hd 720p
- After effects video copilot plugins free download
- The immortals movie free online
- Animated home screen
- Youtube logo maker
- Kruti dev hindi typing finger set
- Microsoft access template for project management
- Krunker io aimbot
- Super smash bros brawl dolphin download
- Traveling budget plan template
- Ip booter free no sign up
- Photoshop cs5 extended free download with crack
- Cakelike oatmeal raisin cookie recipe
- Dfx audio enhancer cracked free download
- Libreoffice dark theme download
- Easy cut studio activation key
- Download logitech gaming software g29
- Sims 4 crack reloaded
- Game revolution sonic mania review
- Credit card validator apk
- Animal revolt battle simulator free game
- Roadblocks video game xbox
- Eset license key generator
- Position chart for trombone
- Extension cord with alligator clips
- Best size for youtube banner
- Leak gta 6 video
- Microsoft word avery template free
- Google earth satellite map
- Silent hunter 4 controls keyboard
- Webstorm 2016-1 license key
- Sakura dungeon porn patch
- Input mapper xinput test not working bluetooth
